News
Gastrulation is one of the most important phases in early embryonic development. Before gastrulation, vertebrate embryos are simple two-dimensional sheets of cells. By the end of gastrulation, an embryo will have begun to differentiate distinct cell types, set up the basic axes of the body and internalize some of the precursors for organs in a three-dimensional structure. Amniotes, like chickens and humans, will have developed a primitive streak, the precursor to the brain and skin, while fish and amphibians will have developed a spherical-shaped blastopore.
Gastrulation is a feat of self-organization, requiring the ballet-like coordinated movements of hundreds to tens of thousands of cells. But, despite its importance in development, scientists only partially understand the underlying mechanisms that coordinate this large-scale movement of cells.
Now a team of researchers from the Harvard, the University of California San Diego and the University of Dundee in the U.K., have developed a theoretical framework that can reproduce and predict the patterns associated with gastrulation in a chicken embryo.
Building on their experimental findings published earlier this year in Science Advances, and using a combination of theory and experiment, the researchers demonstrated that small changes in cell parameters and behavior can have a dramatic impact on the resulting gastrulation patterns.
The new research was also published in Science Advances.
“Linking the developmental processes underlying morphogenesis in an organism to the variations of these same processes across evolution is an old question in biology,” said L. Mahadevan, the Lola England de Valpine Professor of Applied Mathematics at the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS), and Professor of Organismic and Evolutionary Biology, and of Physics in the Faculty of Arts and Sciences (FAS) and senior author of the paper. “Moving away from the gene-centric molecular focus on this question, we asked if there are emergent biophysical principles that operate on the multi-cellular level and can help explain the self-organized gastrulation patterns from both a developmental and an evolutionary perspective.”
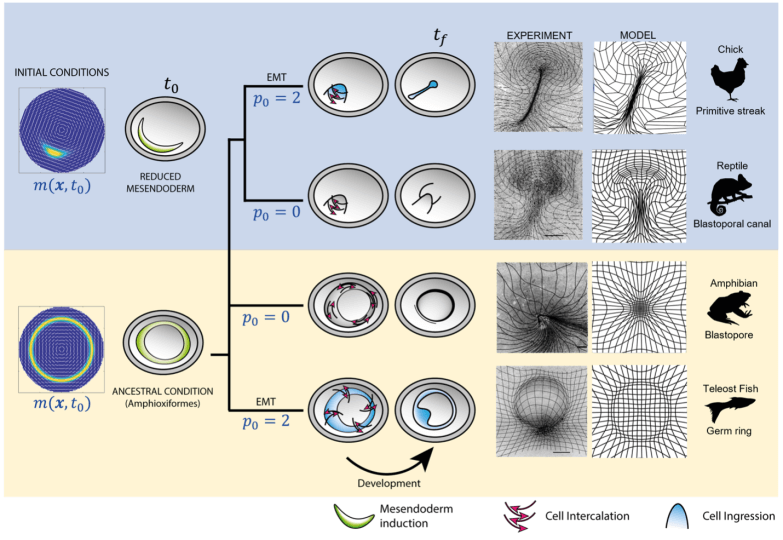
Working closely with experimentalists, the researchers developed a theoretical and computational model that could recreate the movement of the epithelial layer of cells in chick embryos during gastrulation. The team then identified two parameters — one related to the initial distribution of cells in an embryo and the other related to cell behavior — to tweak during gastrulation.
“When we changed these two parameters in the model, which was only informed by the mechanisms of chick development, it remarkably showed gastrulation patterns naturally seen in other species,” said Mattia Serra, first author of the paper and former Schmidt Science fellow in the Mahadevan group.
Serra is an Assistant Professor in Physics at UC San Diego.
Consistent with the computational results, experiments show that perturbing the same parameters in vivo in a chick embryo caused the chick to form a disk-like blastopore, seen in frogs, a ring-like circular primitive streak, like those seen in fish, or an elongated elliptical canal-like streak seen in chameleon.
Changing cell behaviors and initial cell types, (left column), the model recapitulates the flow patterns in the phylogeny of vertebrate gastrulation from a self-organizing dynamical structure. The model-based predicted flow patterns mimic those naturally observed in reptiles, amphibians, and fish, and are reproduced experimentally, in vivo, in the chick embryo. The right columns show deformed Lagrangian grids overlaying the light-sheet microscope images from perturbed chick-experiment velocities and deformed Lagrangian grids from the predicted model velocity for each gastrulation mode. (Credit: Harvard SEAS)
“Our work suggests that the general biophysical principles underlying active self-organized flows and forces during embryogenesis have the power to explain developmental processes and their evolutionary variations across different species of vertebrates,” said Mahadevan.
“We were surprised to see how simple mechanochemical rules could predict highly distinct coherent flows of thousands of cells measured in a living chick embryo," said Serra.
This research not only sheds light on the principles for self- organization in early development but could also help researchers understand the evolutionary history of developmental processes and suggest ways to control the development of synthetic organoids.
This research looked at the early stages of development, when the embryo is just transforming from two-dimensions into three. Next, Mahadevan wants to understand what happens when the embryo starts to fold and buckle to create the more complex shapes of organs and eventually whole organisms.
“Using our knowledge of developmental processes at the molecular and cellular level, we eventually hope to provide an integrated framework for how cells form into tissues and tissues into organs, towards a better understanding of morphogenesis,” said Mahadevan.
This research was co-authored by Guillermo Serrano Nájera, Manli Chuai and Cornelis J. Weijer of the University of Dundee, Alex M. Plum and Sreejith Santhosh of the University of California San Diego and Vamsi Spandan of SEAS. It was supported in part by the Schmidt Science Foundation, the Hellman Foundation, the NSF-Simons Center for Mathematical and Statistical Analysis of Biology under Award 1764269, the National Institutes for Health under grant 1R01HD097068, the Simons Foundation, and the Henri Seydoux Fund.
Cutting-edge science delivered direct to your inbox.
Join the Harvard SEAS mailing list.
Scientist Profiles
L Mahadevan
Lola England de Valpine Professor of Applied Mathematics, of Organismic and Evolutionary Biology, and of Physics
Press Contact
Leah Burrows | 617-496-1351 | lburrows@seas.harvard.edu