News
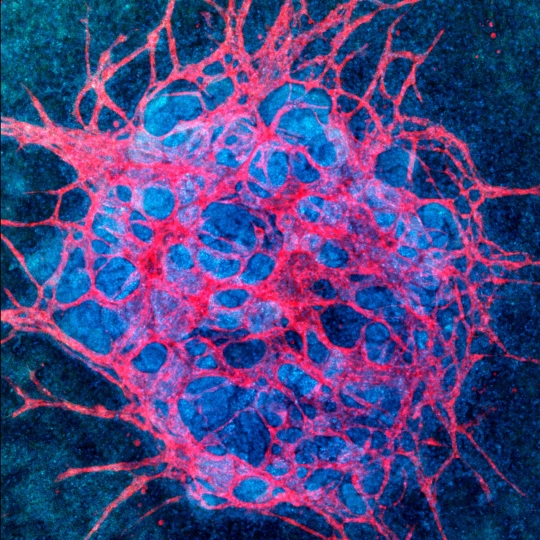
Jennifer Lewis’ and Ryuji Morizane’s teams found previously that culturing kidney organoids under fluid flow causes endogenous endothelial progenitors to create more mature vascular networks which pervade the whole organoid and interact with epithelial compartments. This finding presents a critical advance toward the future development of new approaches by Trestle to treat chronic kidney disease using 3D bioprinting and stem cell-based tissue engineering. Credit: Wyss Institute at Harvard University
A newly launched startup is building upon innovations developed over several years at the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS), the Wyss Institute for Biologically Inspired Engineering at Harvard University, and Brigham and Women’s Hospital (Brigham) to engineer functional kidney tissue for renal repair and replacement therapy.
San Diego-based Trestle Biotherapeutics has been granted a license, by Harvard’s Office of Technology Development (OTD), to commercialize a suite of stem cell- and 3D bioprinting-based kidney regenerative medicine technologies developed at SEAS, Harvard’s Wyss Institute, and Brigham. Founded in 2020, Trestle is led by Ben Shepherd and Alice Chen, both with deep experience in commercial efforts focused in stem cell biology, tissue engineering, regenerative medicine, and 3D bioprinting.
The lack of transplantable kidney tissues and whole organs is an enormous challenge in the treatment of chronic and end-stage kidney disease. As of 2021, in the U.S. alone, there are more than 550,000 dialysis-dependent patients and 100,000 patients waiting for kidney transplants, yet only about 20,000 transplants are performed each year, with nearly 5,000 patients on waiting lists dying each year without receiving a transplant. Beyond kidney failure, there are more than 60 genetic diseases that directly or indirectly affect renal function, many of which cannot be appropriately treated with existing therapeutics.
The foundational technology was pioneered by researchers in the lab of Jennifer Lewis, the Hansjörg Wyss Professor of Biologically Inspired Engineering at SEAS, Wyss Core Faculty member and an Affiliated Faculty member of the Harvard Stem Cell Institute (HSCI). Lewis also leads the Wyss Institute’s Organ Engineering Initiative. Lewis’ lab has collaborated with several clinicians, including Joseph Bonventre, Chief of the Division of Renal Medicine and Founding Chief of the Engineering in Medicine Division at the Brigham, and Ryuji Morizane, who is now an Assistant Professor at the Massachusetts General Hospital (MGH) and Harvard Medical School (HMS), an Affiliated Faculty member at Harvard Stem Cell Institute, and a Visiting Scholar at the Wyss Institute.
“We’re very excited to bring the biofabrication and stem cell biology technologies developed in the Lewis and Morizane labs into what we are building at Trestle. The teams at Harvard, Wyss, and the Brigham have a renowned track record in translational innovation, and we’re thrilled with the opportunity to carry their work forward for patients’ benefit,” said Shepherd, the CEO of Trestle.
“More than a dozen members of my lab contributed to the innovations in tissue engineering that have created this technology platform,” said Lewis. “Most recently, we developed a new biomanufacturing method, known as sacrificial writing in functional tissue (SWIFT), that enables the fabrication of vascularized kidney tissues. I’m pleased that Trestle has now launched to translate this robust technology to address the growing need for kidney tissues and organs.”
Supported in part by Harvard OTD’s Physical Sciences & Engineering Accelerator, and later by the Wyss Institute's Organ Engineering Initiative, Lewis and her colleagues at SEAS and the Wyss made seminal contributions to the field of multimaterial 3D bioprinting. Using their platform technology, Lewis and her collaborators have created 3D kidney-on-chip models for drug screening and disease modeling and have established foundational technology for rapidly generating vascularized kidney tissue at scale for repair and regeneration.
Central to the success of this approach was a collaboration between the Lewis Lab and researchers at the Brigham. Morizane, Bonventre, and other clinicians had developed a method for generating kidney organoids from human pluripotent stem cells in vitro. These tissue constructs contained large numbers of well-organized nephrons, but lacked a vascular network that could be perfused with blood. This deficiency was overcome by subjecting the organoids to the flow of fluids on bioengineered devices. In a collaborative effort published in Nature Methods, the joint team succeeded in generating vascularized kidney organoids with enhanced nephron maturation in vitro.
“We were able to demonstrate for the first time a more advanced kidney architecture and functionality in human kidney organoids, which is important for creating tissue segments for use in drug testing and disease modeling and, ultimately, in vivo therapeutics,” said Morizane. Lewis and Morizane are members of Trestle Biotherapeutics’ scientific advisory board.
By combining these approaches with additional solutions for cell and tissue manufacturing, internal expertise in stem cell and kidney organoid biology, and the commercialization of 3D bioprinted human tissues, the team at Trestle will work towards its goal of creating a new standard-of-care option for patients with kidney failure through the development of bioengineered kidney tissue with the ability to assume vital kidney functions.
Topics: Bioengineering, Technology
Cutting-edge science delivered direct to your inbox.
Join the Harvard SEAS mailing list.
Scientist Profiles
Jennifer Lewis
Hansjorg Wyss Professor of Biologically Inspired Engineering
Press Contact
Leah Burrows | 617-496-1351 | lburrows@seas.harvard.edu