News
Key Takeaways
- A new Harvard study describes a way to process natural rubber that preserves its long molecular chains while mixing in strength-endowing particles.
- The resulting stiff and durable material could lead to strong, long-lasting, natural-rubber composite tires that reduce waste and dust pollution.
Natural rubber, tapped from trees as latex, is the world’s most widely used bio-elastomer. Comprising long molecular chains that make it pliable and stretchy yet highly resistant to cracking and strain, natural rubber is foundational to countless products, including the heavy-duty tires in trucks, buses, and airplanes.
But to make rubber products that can withstand those demanding applications, large machines must mix in reinforcing particles, usually carbon black or silica, that chew up the rubber’s long polymer chains and effectively sacrifice many of the natural material’s most exceptional properties, from elasticity to cold resistance. The result is a tire that’s sufficiently stiff, yet prone to wearing and cracking, which produces polluting dust as it wears and decreases its lifespan.
It doesn’t have to be this way, says a new Harvard study. A team from the lab of Zhigang Suo, the Allen E. and Marilyn M. Puckett Professor of Mechanics and Materials in the John A. Paulson School of Engineering and Applied Sciences (SEAS), along with Yakov Kutsovsky, Expert-in-Residence at Harvard’s Office of Technology Development, has developed a simple new way to process natural rubber that preserves its unique long molecular chains while uniformly mixing in strength-endowing particles. The result is a composite material that is both unusually stiff and extraordinarily tough – properties that rarely go hand in hand in rubber products.
The work, published in Proceedings of the National Academy of Sciences, points to a future where more durable truck, bus and airplane tires can be made from tree-derived rubber, with potential to cut waste, reduce tire dust pollution, and open new avenues for high-performance elastomers in applications such as soft robotics. It also seeds an eventual pathway to upending the industry for synthetic rubber tires, which are used in passenger cars and derived from petroleum.
“Waste can come in the form of these tire dust particles, and it can come in the form of just throwing tires away,” said first author Matthew Wei Ming Tan, former Harvard postdoctoral researcher and now an assistant professor at Nanyang Technological University, Singapore. “We eventually want a circular economy that minimizes waste and cycles materials back into use. Extending tire lifespan is a key part of that — the longer each tire lasts, the fewer raw materials we need to extract and the less waste we generate in the first place.”
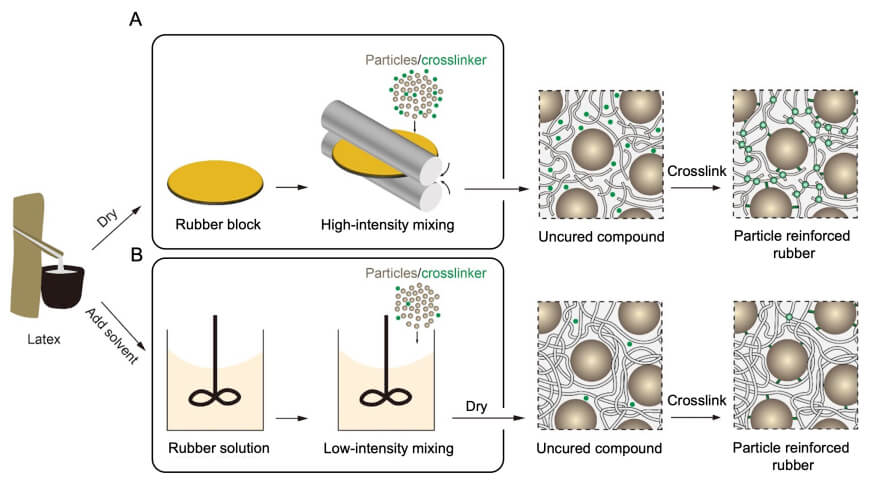
Until now, the challenge has been how to mix solid particles into natural rubber without tearing those long chains apart. In latex, natural rubber exists as tiny spheres — “like meatballs in water,” as Tan describes it. Simply adding particles to that latex doesn’t work: the particles stay in the water and fail to penetrate the rubber spheres. Dry‑mixing, as used today, solves the mixing problem but at the cost of cutting that chains, known as chain scission.
Solution-based mixing process
In the new work, the researchers first transformed those rubber spheres into a solution of individual chains. They used a common organic solvent, toluene, whose polarity matches that of the rubber, allowing the long chains to dissolve out of the spherical particles and into a uniform “rubber soup.” Silica powder is then blended into this solution, which is later dried to recover a solid compound.
After drying, the material can be stored, molded into shape, and then cured. During curing, the long chains are only sparsely cross‑linked with each other and interlinked with the silica particles.
Tests show that preserving the long chains while adding silica dramatically boosts performance. The toughness — a measure of how much energy a material can absorb before a crack grows — rose by about an order of magnitude, from roughly 2 to 44 kilojoules per square meter. At the same time, the material maintained a high modulus (stiffness) of about 19 megapascals.
Silica-reinforced natural rubber processed in two routes, with and without mastication. A conventional route begins by mixing dry rubber, silica particles, and additives by high-intensity processes, which masticate polymer chains. A new solution-based process begins with dissolving latex particles into rubber chains in toluene. The solution is mixed with silica particles by low-intensity mixing, which retains long chains.
Tire wear is an emerging environmental concern. As treads abrade, they release fine dust that contains rubber fragments and various additives. These microscopic particles accumulate in air, soil, rivers, and oceans, where studies have linked some additives to harmful effects on fish and potentially on human health.
While the new study focuses on fundamental fracture and fatigue properties rather than direct abrasion tests, improved crack resistance is generally understood to correlate with better wear resistance.
Tires, belts, robotic components
Although the immediate relevance is to large truck, bus, and aircraft tires that already rely on natural rubber, the researchers see broader possibilities. If costs and supply can be managed, similar approaches could one day enable high‑performance passenger‑car tires made largely from bio‑based rubber rather than oil‑derived synthetics. Beyond tires, any application that demands repeated flexing — industrial belts, seals, and especially soft robotic components — could benefit.
The rise of electric cars makes the advances even more pressing, said co-author Kutsovsky, who previously served as Chief Scientific Officer and Chief Technology Officer at Cabot Corporation developing materials for elastomer reinforcements in tires and other applications. “Electric cars are heavier than ordinary cars, and larger loads on the car is going to make the tires wear out faster,” Kutsovsky said. “So while the engine and batteries are quite efficient, the tires become a bigger component of energy loss.”
The solvent used in the current experiments is widely used in industry and can be handled in closed‑loop, fully recyclable systems. Tan and Kutsovsky note that the overall processing strategy is general and has already been demonstrated with both silica and carbon black, as well as with different grades of silica.
“Amplifying toughness in silica-reinforced natural rubber by preserving long chains” is co-authored by Guodong Nian, Zheqi Chen, and Xianyang Bao. Federal support for the work came from the National Science Foundation under MRSEC (DMR-2011754), and the Air Force Office of Scientific Research under award No. FA9550-20-1-0397. The work was performed in part at the Harvard University Center for Nanoscale Systems, a member of the National Nanotechnology Coordinated Infrastructure Network, which is supported by the NSF under award No. ECCS-2025158.
Topics: Industry, Materials, Materials Science & Mechanical Engineering, Research, Robotics, Technology
Cutting-edge science delivered direct to your inbox.
Join the Harvard SEAS mailing list.
Scientist Profiles
Zhigang Suo
Allen E. and Marilyn M. Puckett Professor of Mechanics and Materials
Press Contact
Anne J. Manning | amanning@seas.harvard.edu